What is RegenKit®BCT ?
The RegenKit® BCT are medical devices intended for the preparation of RegenPRP.
They contain RegenBCT® tubes and accessories for blood collection and PRP recovery. RegenBCT® tubes are made of pharmaceutical grade glass with a vacuum for automated blood collection.
They contain a sodium citrate anticoagulant solution and a separating gel to separate plasma and platelets from the blood cells and produce RegenPRP® with a standardized composition.
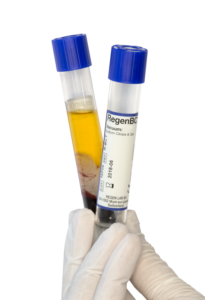
Technology platform for standardized autologous regenerative medicine
The simple, safe and efficient point-of-care preparation of autologous platelet-rich plasma.
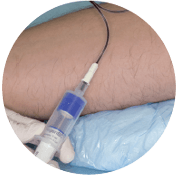 Blood Collection
Blood Collection
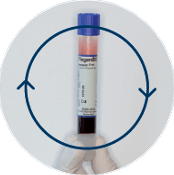 Centrifugation
Centrifugation
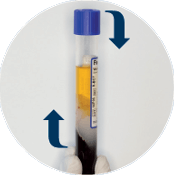 Platelet Resuspension
Platelet Resuspension
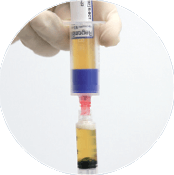 Ready to use
Ready to use
TECHNOLOGY ADVANTAGES
- User-independent standardized preparation
- Minimum volume of blood required
- Safe closed-circuit system
- Mechanical isolation of PRP using a biologically inert gel after a 5-minute centrifugation
- Reversible anticoagulation with a pharmaceutical grade solution of sodium citrate at pH 7
- Minimal learning curve and ease of use
- Operationally and clinically efficient process
- Facilitates and streamlines routine practice
Scientific advantages
- Demonstrated safety and efficacy
- Evidence-based outcomes for numerous therapeutic indications
- Large number of clinical studies, with over 200 publications
Biological advantages
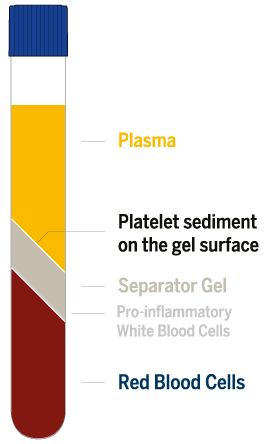
- RegenPRP® is standardized, leucocyte reduced and easily reproducible
Regen Lab specific separating gel technology guarantees minimal variability - Platelet recovery >80%
- High platelet quality
Viable & functional platelets - Full plasma recovery
No loss of plasma growth factors and fibrinogen - Leucocyte reduced PRP
Depletion ~ 96.7% of pro-inflammatory granulocytes, leaving mainly lymphocytes and monocytes - Virtually no red blood cells
Depletion of ~ 99.7% of erythrocytes
RegenPRP® standardized performance
Regen Lab devices use a separating gel technology that separates the plasma and
platelets from the blood cells to produce RegenPRP with a standardized composition:
RegenBCT® Tube Properties

Blood sample vol per tube
10 ml

PRP vol per tube
5 to 6 ml

Platelet recovery
> 80%

Red blood cell depletion
> 99.7%

Platelet concentration factor (native)
1.6 X
Our products
Clinical evidence in knee-osteoarthritis
Intended use of the device
Preparation of autologous platelet-rich plasma (A-PRP®) and other plasma-derived products.
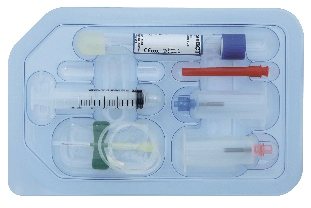

RegenKit®-BCT-1-2
Ref: RK-BCT-1 // RK-BCT-2
1 Safety–Lok™ Butterfly needle
1 Collection holder
1 to 2 Regen BCT* tubes
1 Transfer device
1 to 2 transfer needles
1 to 2 x 5 ml Luer-Lok™ syringe
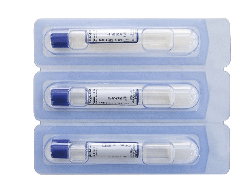

RegenKit® str-BCT-3
Ref: RK-BCT-3
3 Regen BCT* tubes


RegenKit® BCT-T
RK-BCT-T
1 Regen BCT* tube
*BCT stands for Blood Cell Therapy
Warnings and precautions
Strict aseptic technique must be followed during the whole procedure. Use proper safety precautions to avoid contact with patient blood or cross-contamination. Use proper safety precautions to protect against needles or broken tubes. Do not use sterile component of this kit if it is opened or damaged. Do not use components of this kit if they are broken or present a defect. Do not use the tube if it has lost vacuum. Do not use the sodium citrate solution or other tube components separately. Store between 5 ºC and 30ºC; bring the kit to ambient temperature before using tubes. Do not re-sterilise, do not use after the expiration date. Single use device, do not reuse any part of the kit. Reuse may lead to infection or other illness / injury. Transfer needle must be used to transfer liquids only and should not be used for injection. The preparation of the platelet-rich plasma (PRP) must be performed by a physician trained on the equipment and procedure, or under the supervision of the physician. The treatment with PRP must be performed by a qualified physician. Do not inject PRP intravascularly. The patient must be informed of the general risks associated with the treatment and of the possible adverse effects. The safety and effectiveness of combination of PRP with other therapies should be assessed by the physician. The safety and effectiveness have not been evaluated in children and in pregnant or lactating women. The PRP must be prepared from fresh blood and must be used within four hours (extemporaneous use only).
All tubes and components of the kit are to be entirely discarded by elimination method after each use to avoid potential contamination with blood products. Use a 45° fixed angle rotor centrifuge or a horizontal head swinging bucket centrifuge (ex. RegenPRP Centri provided by Regen Lab). Follow the manufacturer’s instructions when using the centrifuge. Tubes should be centrifuged, as recommended in the instructions for use, at a relative centrifugation force (RCF) of 1500 g. Excessive RCF (over 2200 g) may lead to tube breakage resulting in blood exposure, and possible injury. RCF below 1500 g may lead to incorrect blood separation and red blood cell contamination of PRP. Centrifuge carriers and inserts size should be adapted to the tubes. Use of carriers too large or too small may result in breakage of the tubes. Caution should be taken to ensure that tubes are properly seated in the centrifuge carriers. Tubes must be balanced in the centrifuge.
Possible adverse effects
Possible side effects of blood collection
Blood collection may cause damage of the blood vessels, hematomas, superficial phlebitis, delayed wound healing, early or late infection and/or temporary or permanent nerve damage that may result in pain or numbness.
Patented by Regen Lab SA
Platelet Rich Plasma
U.S. patent US8529957, US11241458, US11110128, US11096966, US10881691,
US10092598, US10080770, US10064894, US8529957, US9833478, US10052349, US11241458
European patent EP3111974B1
Swiss patent CH696752
Hong Kong patent HK1231793
Contact Regenlab
Now is the time to change your patient’s life. Browse ressources and
get connected to treatment that is right for them.
For more information on our products, please contact us by clicking on the button below:
Our Regional offices are
located in
- New York (USA)
- Montréal (Canada)
- Venice (Italy)
- Munich (Germany)
- Paris (France)
- Dubai (U.A.E.)
- Beijing (China)
- Istanbul (Turkey)




